Four procedures of Ministry of Health to connect to National Single Window
 |
The Ministry of Health issued a decision on the connection of four administrative procedures related to traditional medicine and pharmacy to the National Single Window across the country.
According to the Ministry of Health, four procedures include issuing a certificate of sale for imported traditional medicine exempted from clinical trial or clinical trial stage exemption; application for re-export of recalled traditional medicine; allowing the import of medicine and medicinal ingredients with the remaining expiry date at the customs clearance time shorter than prescribed; and license for export of herbal ingredients under the list of precious, rare and endemic species and categories subject to control.
The results of these procedures are electronic licenses or certificates which are issued by competent persons with digital signatures of competent agencies.
The Administration of Traditional Medicine and Pharmacy, the Ministry of Health, the General Department of Vietnam Customs and the customs departments are in charge of carrying out these administrative procedures.
Traditional medicine manufactures and importers are allowed to implement these procedures via the National Single Window.
The pilot connection of these procedures to the National Single Window is from December 20, 2021 to January 20, 2022.
The four procedures will be officially connected to the National Single Window from January 21, 2022.
Related News

Customs sector strengthens anti-smuggling for e-commerce products
19:09 | 21/12/2024 Anti-Smuggling
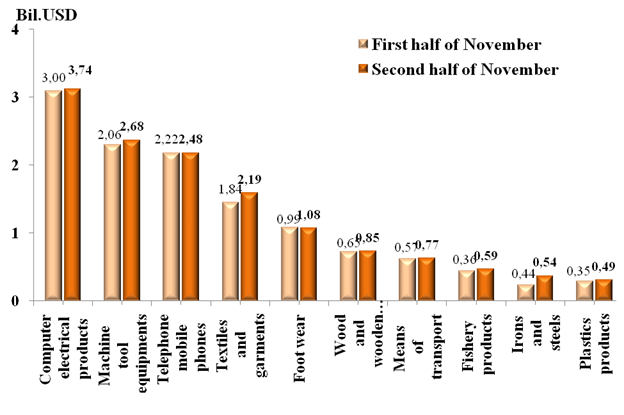
Preliminary assessment of Vietnam international merchandise trade performance in the second half of November, 2024
15:18 | 19/12/2024 Customs Statistics

Achievements in revenue collection are a premise for breakthroughs in 2025
09:57 | 18/12/2024 Customs

Numerous FDI enterprises face suspension of customs procedures due to tax debt
09:57 | 18/12/2024 Anti-Smuggling
Latest News

Minister of Finance Nguyen Van Thang: Facilitating trade, ensuring national security, and preventing budget losses
19:09 | 21/12/2024 Customs

Official implementation of the program encouraging enterprises to voluntarily comply with Customs Laws
18:31 | 21/12/2024 Customs

Proactive plan to meet customs management requirements at Long Thanh International Airport
18:30 | 21/12/2024 Customs

An Giang Customs issues many notes to help businesses improve compliance
09:29 | 20/12/2024 Customs
More News

Hai Phong Customs processes over 250,000 declarations in November
15:18 | 19/12/2024 Customs

Binh Duong Customs surpasses budget revenue target by over VND16.8 Trillion
09:39 | 18/12/2024 Customs

Director General Nguyen Van Tho: Customs sector strives to excellently complete 2025 tasks
16:55 | 17/12/2024 Customs

Customs sector deploys work in 2025
16:43 | 17/12/2024 Customs

Mong Cai Border Gate Customs Branch makes great effort in performing work
11:23 | 16/12/2024 Customs

Declarations and turnover of imported and exported goods processed by Lao Bao Customs surge
09:17 | 15/12/2024 Customs

General Department of Vietnam Customs prepares for organizational restructuring
19:28 | 14/12/2024 Customs

Revenue faces short-term difficulties but will be more sustainable when implementing FTA
19:27 | 14/12/2024 Customs

Customs sector collects over VND384 trillion in revenue
17:13 | 12/12/2024 Customs
Your care

Minister of Finance Nguyen Van Thang: Facilitating trade, ensuring national security, and preventing budget losses
19:09 | 21/12/2024 Customs

Official implementation of the program encouraging enterprises to voluntarily comply with Customs Laws
18:31 | 21/12/2024 Customs

Proactive plan to meet customs management requirements at Long Thanh International Airport
18:30 | 21/12/2024 Customs

An Giang Customs issues many notes to help businesses improve compliance
09:29 | 20/12/2024 Customs

Hai Phong Customs processes over 250,000 declarations in November
15:18 | 19/12/2024 Customs




